Best Practice Updates
Blister packs are being withdrawn – what’s really happening?

Blister packs are being withdrawn – what’s really happening?
You may have noticed that it’s difficult to obtain medicines in monitored dosage systems (MDS or ‘blister packs’ as they are commonly known). These are hard plastic or disposable trays containing a week’s worth of medicines.
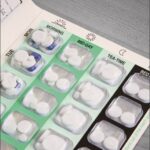
It’s been a few years (January 2019) since Boots first announced they were no longer supply blister packs (or as we pharmacists call them – monitored dosage systems) to care homes: both new and existing ones.
Lloyds pharmacy followed by saying they’ll no longer supply blister packs to new care homes and they’ll review supply to existing care homes on a “case by case” basis. When we asked, we didn’t hear back from any of the other big chains such as Well pharmacy.
None of the pharmacy groups stated if they’re changing the supply of blister packs to people in their own homes. So, although there are no changes due imminently, but it’s case of “watch this space”.
If you find this article useful, sign up for updates when new articles are published (on the left hand side or bottom of the screen for mobiles).
What’s prompted this?
The official line from Boots was that their decision was in response to Royal Pharmaceutical Society (RPS) guidelines (1, 2) and NICE guidelines (3, 4).
The RPS guidelines were issued in 2012 and 2013 (1, 2). They recommended original packs (boxes and bottles) as first choice for all settings, saying blister packs should only be used if there’s a “specific need” which is assessed by a health care professional.
What is wrong with blister packs?
Although they may make administration seem easier, there are risks with blister packs which the RPS reports highlight. This has also been our experience. When we run training sessions we often ask how many people have received blister packs with mistakes in and there is always a strong show of hands. The problem is they are hard to check once they are sealed, so the checking pharmacist can miss mistakes, as can the care workers when they administer from them. Hence, any extra/missing or incorrect tablets in the pack often go straight through and are taken by the client.
What did NICE say?
Not an awful lot actually. In their care homes guidelines (3) NICE said:
- Care home providers should determine the best system based on the individuals needs of the resident
In their community guidance in 2017 (4) NICE basically supported RPS, agreeing that blister packs should only be used when a specific need has been identified by a health professional.
So, the pharmacies are stating this is about NICE guidelines, and NICE are pointing to the RPS guidelines which are around 7 years old. So if the pharmacies have known about this for 7 years, why the sudden change?
What’s really going on?
It’s true that patient safety plays a role, but there is something else: cost.
Pharmacy’s are businesses, with an NHS contract. For each item (medicine) on a prescription they dispense, they claim back the cost of the drug from the government, and then get paid just £1.26 per item on top of that.
Out of these £1.26 pence payments, the pharmacy must pay for the pharmacy technician, the pharmacist, and all the other overheads of running a business on the high street: rent, rates, bills etc.
Now, to dispense into a blister pack (and check it properly) takes time (sometimes up to 45 minutes). That’s significantly longer than attaching a pharmacy label to a manufacturer’s pack. Staff time costs money (pharmacist and dispensing technician costs). Add to that the cost of the blister pack (about 30 pence), and the pharmacy are probably losing money dispensing into blister packs.
Now add in austerity. In the last few years, pharmacies have suffered a real reduction in what they get paid by the government. So much so, Lloyds and Boots are closing hundreds of branches. So this isn’t a good time to provide a service that loses you money!
And there is something ….. eMAR
eMAR stands for electronic MAR (medicines administration record) charts. Most eMAR systems are designed to be used with manufacturer’s original packs, not blister packs. Hence pharmacies have seen this as an opportunity to encourage care homes away from blister packs and back into original manufacturers’ packs.
However blister packs are not disappearing altogether. There are some pharmacies that are still providing these to care homes, and most pharmacies are still providing these to people in their own homes (as long as there is a genuine need for them). But quite how long that continues in the face of cost pressures remains to be seen…
What does this mean for you and the client?
If you have a client who genuinely needs a blister pack, talk to the pharmacist, as under the Equality Act 2010, the pharmacy have an obligation to make what are called ‘reasonable adjustments’ to their service (i.e. supply in blister packs if that is appropriate). However, it’s about the client’s needs, not the care service’s needs to have a quick and convenient method for staff to administer medicines from.
Click here to see our Foundation Courses in Medicines Administration. They teach how to administer from blister packs as well as from original packs.
John Greene MRPharmS and Susie Ogden MPharm
References:
(1) Improving pharmaceutical care in care homes . Royal Pharmaceutical Society March 2012
(2) Improving patient outcome. The better use of multi-compartment compliance aids. Royal Pharmaceutical Society July 2013
(3) Managing medicines in care homes. NICE guidelines March 2014
(4) Managing medicines for adults receiving social care in the community. NICE guidelines March 2017
